News

05 June 2026
Upcoming DBT sessions will take place on 26 June and 8 July 2026.
Details
News

04 June 2026
DBT is organising a two-part webinar series designed specifically for UK SMEs on 5 and 19 June 2026.
Details
Press

02 June 2026
CTPA with Forum for the Future launches "Navigating a Volatile World – A Handbook for the UK Cosmetics Industry".
Details
News

02 June 2026
CTPA with Forum for the Future launches "Navigating a Volatile World – A Handbook for the UK Cosmetics Industry".
Details
News

28 May 2026
The Society of Cosmetic Scientists have unveiled a refreshed identity, including a new name, logo and digital presence
Details
News

28 May 2026
TFWA World Exhibition & Conference will be held from 27 September to 1 October 2026.
Details
News

22 May 2026
The deadline for responses is 10 August 2026.
Details
News

21 May 2026
The UK-GCC Free Trade Agreement was concluded on 20 May 2026.
Details
News

19 May 2026
Webinar on 2 June 2026 - Apprenticeship scheme launched in partnership with SCS and Tiro
Details
News

14 May 2026
Join DBT on 28 May 2026 to prepare for the upcoming India-UK FTA Roadshow!
Details
News

13 May 2026
Upheld ruling on misleading claims which exaggerated the effects of a cosmetic for ageing skin.
Details
Press

News

13 May 2026
The SCS 2026 Annual Conference is taking place on 1-2 July 2026
Details
News

08 May 2026
The recording of the ICCS webinar on skin sensitisation is now available.
Details
News

08 May 2026
The HSE published an SI updating the provisions of the GB CLP Regulation.
Details
News

05 May 2026
Glass containers from China and Turkey are under investigation. Survey closes on 21 May 2026.
Details
News

30 April 2026
Join DBT's webinar with HM Trade Commissioner on 5 May 2026 at 11am.
Details
News

30 April 2026
Upheld ruling on misleading clinically proven claim to look up to five years younger.
Details
News
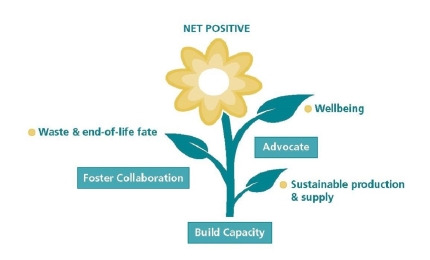
30 April 2026
Join CTPA’s webinar for a discussion around the tools that will help you and your team spark insight and navigate a volatile world.
Details
News
28 April 2026
Roadshow events will take place between 17-25 June 2026 across several UK locations.
Details
News

28 April 2026
DBT is organising a pitching day where UK Businesses can pitch directly to buyers in India.
Details
News

27 April 2026
Latest insights from Dr Emma Meredith OBE, Director-General, CTPA
Details